
Technical Cleanliness
Technical cleanliness is critical for applications in sensitive industries such as electronics or medical technology. Krückemeyer manufactures adhesive solutions under controlled conditions and offers materials- and process-appropriate products in accordance with the highest cleanliness requirements.


Cleanroom production
Controlled cleanliness for sensitive processes: In the Cleanroom production, adhesive tapes, films and foams are processed under strictly defined conditions – ideal for medical, pharmaceutical, electronics and optical industries, where particle-free conditions and process reliability are crucial.
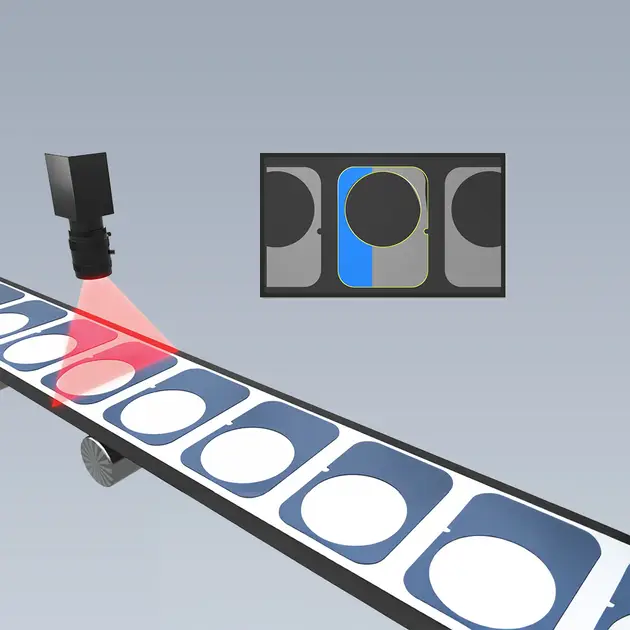
Optical Metrology
Precision down to the last detail: The optical metrology ensures that adhesive parts, film cuts, and stamped parts conform exactly to the required dimensions and tolerances — for high fit accuracy, reproducible quality, and stable processes in mass production.
Technische Sauberkeit in der Klebtechnik
Technical cleanliness means: Particles, fibers, residues and contaminants are consistently minimized – so that adhesive joints bond reliably and perform, look visually clean, and can be processed in both manual and automated processes without disruption.
Technische Sauberkeit
I - Cleanroom ISO 9
When applications require the highest level of cleanliness, manufacturing takes place under defined cleanliness or cleanroom conditions. This keeps adhesive parts free from dust, fibers and foreign particles – from manufacturing to packaging.
II - Stamping precision
Clean stamping contours are a cleanliness factor: exact geometries, smooth edges and defined tolerances prevent material wear, fraying and particle formation – and ensure process-safe dispensing.
III - Cutting precision
Clean, precise cutting provides dimensionally accurate widths, straight edges and accordingly clean winding patterns. This reduces edge wear, minimizes disturbances during unwinding/dispensing and supports clean, automatable processes.
Absolut reproduzierbar
I - Fully reproducible product qualities
Fully reproducible product qualities mean consistent results in the process: less downtime, less scrap, less rework—and reliable batch quality.
II - Optical metrology using a line-scan camera
For technical cleanliness and dimensional accuracy: With optical inspection (e.g., a line-scan camera), stamped parts are measured automatically. Process metrics (e.g., CpK values) make the stability visible and verifiable.
III - Quality assurance documentation
Traceability requires documentation: a complete QA documentation with test protocols, batch traceability and release approvals ensures that quality is not only achieved but permanently safeguarded.

Ihr Ansprechpartner
Thomas Klein
- Expert for customized adhesive solutions
- Project business “Klebtechnik”
- For over 20 years in the industry





